Quantifying and modelling the acquisition and retention of lumpy skin disease virus by haematophagus insects reveals clinically but not subclinically-affected cattle are promoters of viral transmission and key targets for control of disease outbreaks
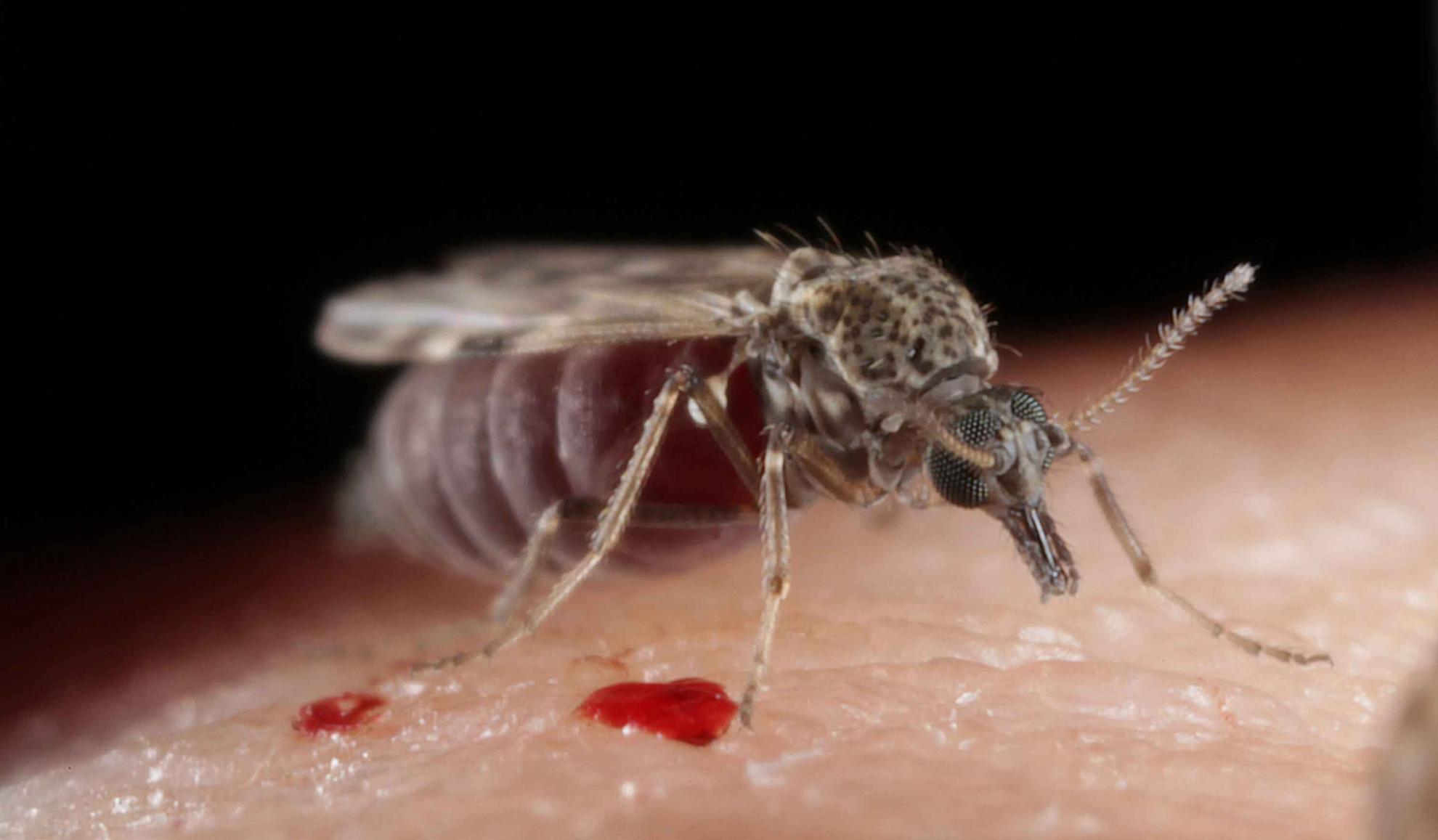
Lumpy skin disease virus (LSDV), is a vector-transmitted poxvirus that causes disease in cattle. The vector species involved in LSDV transmission and their ability to acquire and transmit the virus are poorly characterised. Using a highly representative bovine experimental model of lumpy skin disease we fed four model vector species (Aedes aegypti, Culex quinquefasciatus, Stomoxys calcitrans and Culicoides nubeculosus) on LSDV-inoculated cattle in order to examine the acquisition and retention of LSDV by these species. We found the probability of LSDV transmission from clinical cattle to vector correlated with disease severity. Subclinical disease was more common than clinical disease in the inoculated cattle, however the probability of vectors acquiring LSDV from subclinical animals was very low.
All four potential vector species studied had a similar rate of acquisition of LSDV after feeding on the host, but Aedes aegypti and Stomoxys calcitrans retained the virus for a longer time, up to 8 days. There was no evidence of virus replication in the vector, consistent with mechanical rather than biological transmission. The parameters obtained in this study were combined with data from previously published studies of LSDV transmission and vector life history parameters to determine the basic reproduction number of LSDV in cattle mediated by each of the model species. This was highest for Stomoxys calcitrans (19.1), C. nubeculosus (7.4), and Ae. aegypti (2.4), indicating these three species are potentially efficient transmitters of LSDV, which can be used to inform LSD control programmes.
Importance: Lumpy skin disease virus (LSDV) causes a severe systemic disease characterised by cutaneous nodules in cattle. LSDV is a rapidly emerging pathogen, having spread since 2012 from Africa and the Middle East into Europe, Russia, and across Asia. The vector-borne nature of LSDV transmission is believed to have promoted the rapid geographic spread of the virus, however a lack of quantitative evidence about LSDV transmission has hampered effective control of the disease during the current epidemic. Our research has combined experimental and modelling approaches in order to calculate the reproductive number of different insect species, therefore identifying efficient transmitters of LSDV. It has also characterised a subclinical form of LSDV in cattle and shown that these animals play little part in virus transmission. This information can be used to devise evidence-based, proportionate, and effective control programmes for LSD.