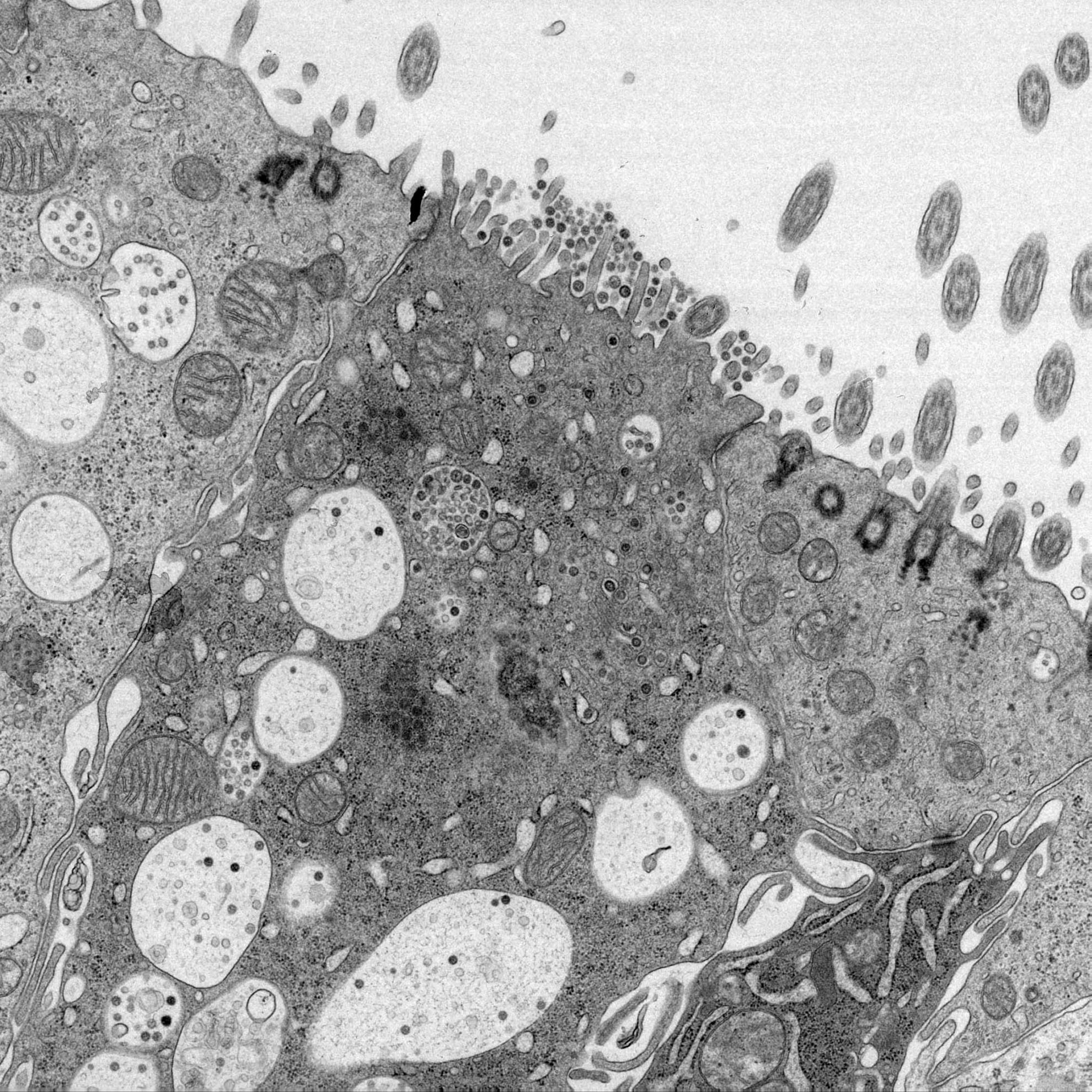
Coronavirus nsp6 proteins generate autophagosomes from the endoplasmic reticulum via an omegasome intermediate
Autophagy is a cellular response to starvation which generates autophagosomes to carry cellular organelles and long-lived proteins to lysosomes for degradation. Degradation through autophagy can provide an innate defense against virus infection, or conversely autophagosomes can promote infection by facilitating assembly of replicase proteins. We demonstrate that the avian coronavirus, infectious bronchitis virus (IBV), activates autophagy. A screen of individual IBV nonstructural proteins (nsps) showed that autophagy was activated by IBV nsp6. This property was shared with nsp6 of mammalian coronaviruses mouse hepatitis virus, and severe acute respiratory syndrome virus, and the equivalent nsp5-7 of the arterivirus porcine reproductive and respiratory syndrome virus. These multiple-spanning transmembrane proteins located to the endoplasmic reticulum (ER) where they generated Atg5 and LC3II-positive vesicles, and vesicle formation was dependent on Atg5 and class III PI3 kinase. The vesicles recruited double-FYVE-domain containing protein (DFCP) indicating localized concentration of phosphatidylinositol 3 phosphate, and therefore shared many features with omegasomes formed from the ER in response to starvation. Omegasomes induced by viral nsp6 matured into autophagosomes that delivered LC3 to lysosomes and therefore recruited and recycled the proteins needed for autophagosome nucleation, expansion, cellular trafficking and delivery of cargo to lysosomes. The coronavirus nsp6 proteins activated omegasome and autophagosome formation independently of starvation, but activation did not involve direct inhibition of mTOR signaling, activation of sirtuin 1 or induction of ER stress.
Back to publications