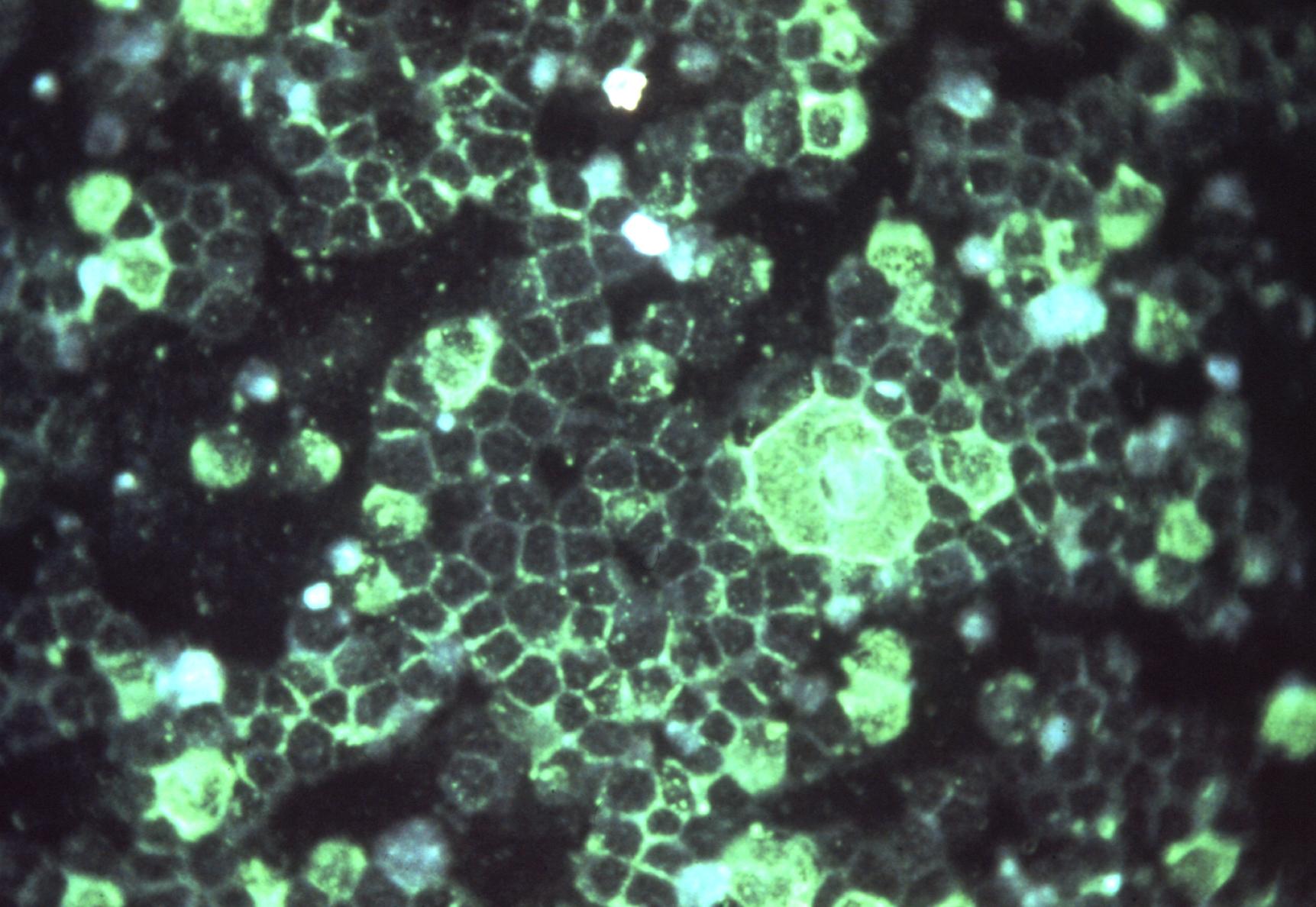
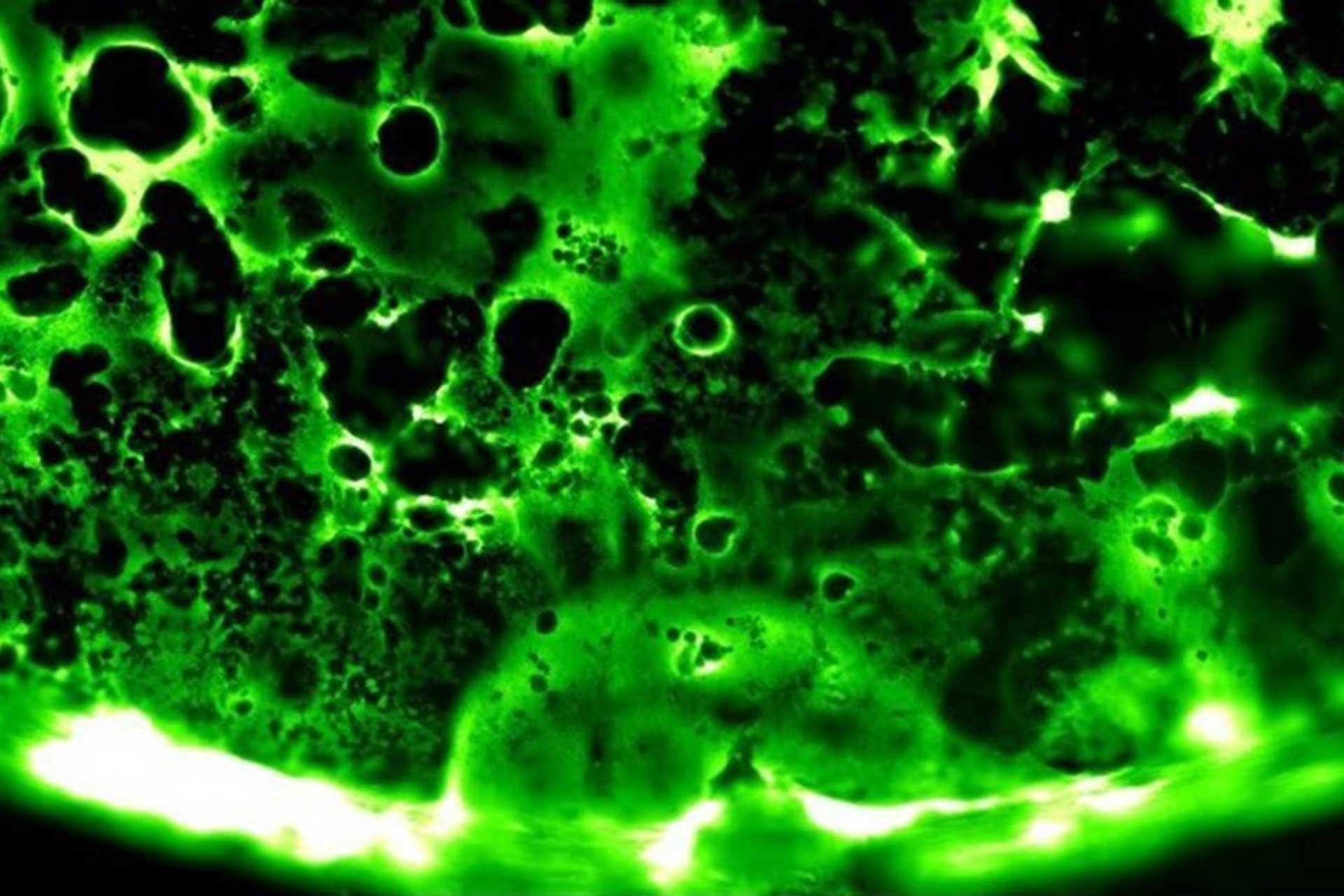
Human respiratory syncytial virus (RSV) infects the lower respiratory tract of humans, causing the fusion of infected cells into syncytia (hence the virus name).
The virus is spread by direct contact with infected secretions and transmission through objects (fomites).
RSV is prevalent worldwide, with nearly all humans being infected by the age of two. Most infections result in mild, cold-like symptoms, but 1-2% of cases can progress to more severe conditions such as bronchiolitis or pneumonia, often necessitating hospitalisation.
Repeated infections can occur in immunocompromised adults and in the elderly.
Annual epidemics occur during the winter months in temperate climates and during the rainy season in tropical climates.
Clinical signs
RSV causes infections mainly during infancy and childhood, although infections can occur in the elderly and adults that have weakend immune systems.
Clinical Signs:
- Runny nose
- Decrease in appetite
- Coughing
- Sneezing
- Fever
- Wheezing
- Irritability
- Decreased activity
- Breathing difficulties
- Bronchitis
- In severe cases death
Pirbright's research on respiratory syncytial virus
RSV causes 200,000 deaths worldwide per year in children under the age of five and there is currently no vaccine. However, work done at the Institute has shown a new type of vectored vaccine provides immunity against bovine respiratory syncytial virus, a closely related virus in calves, giving strong evidence that a similar type of vaccine could work in humans.