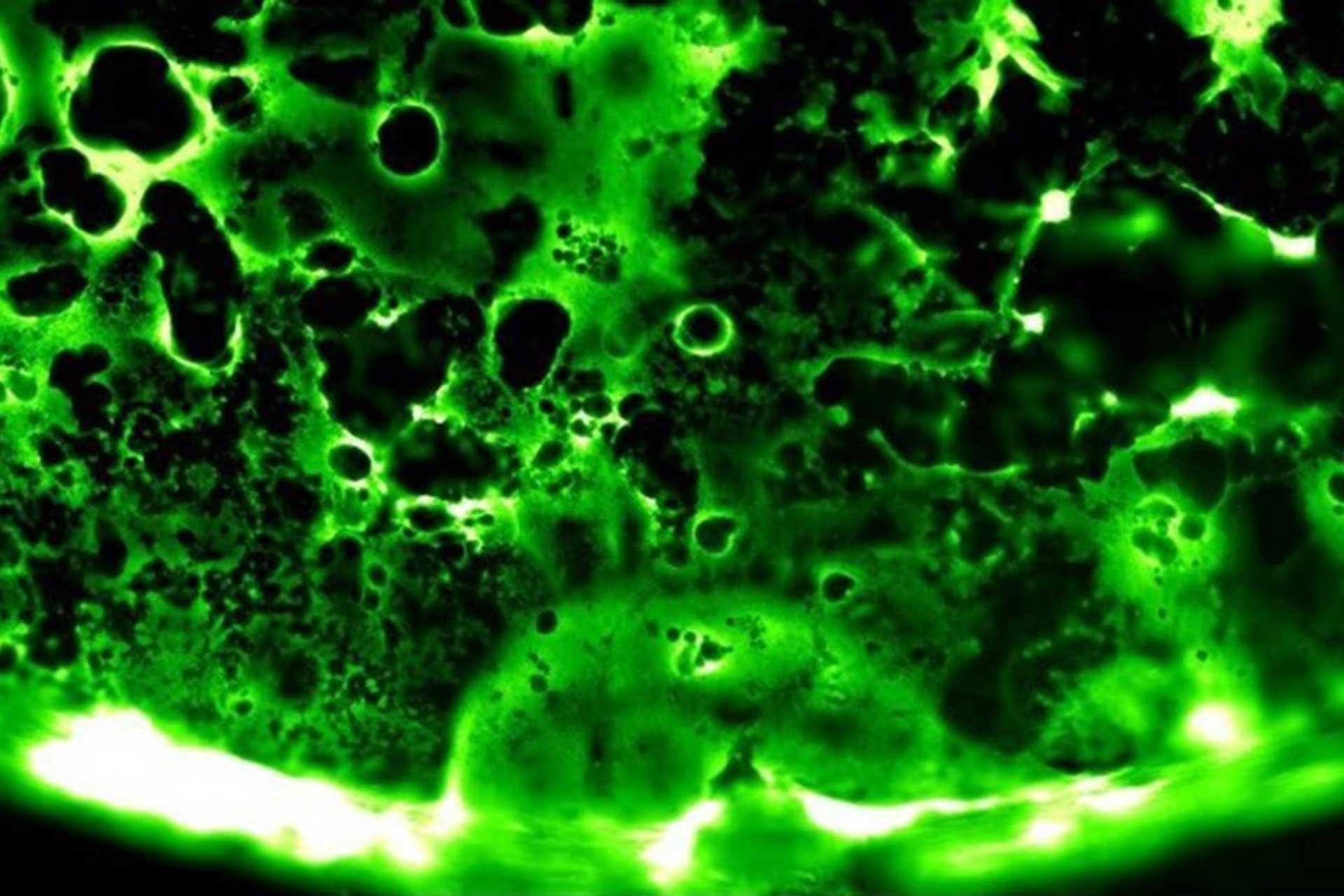
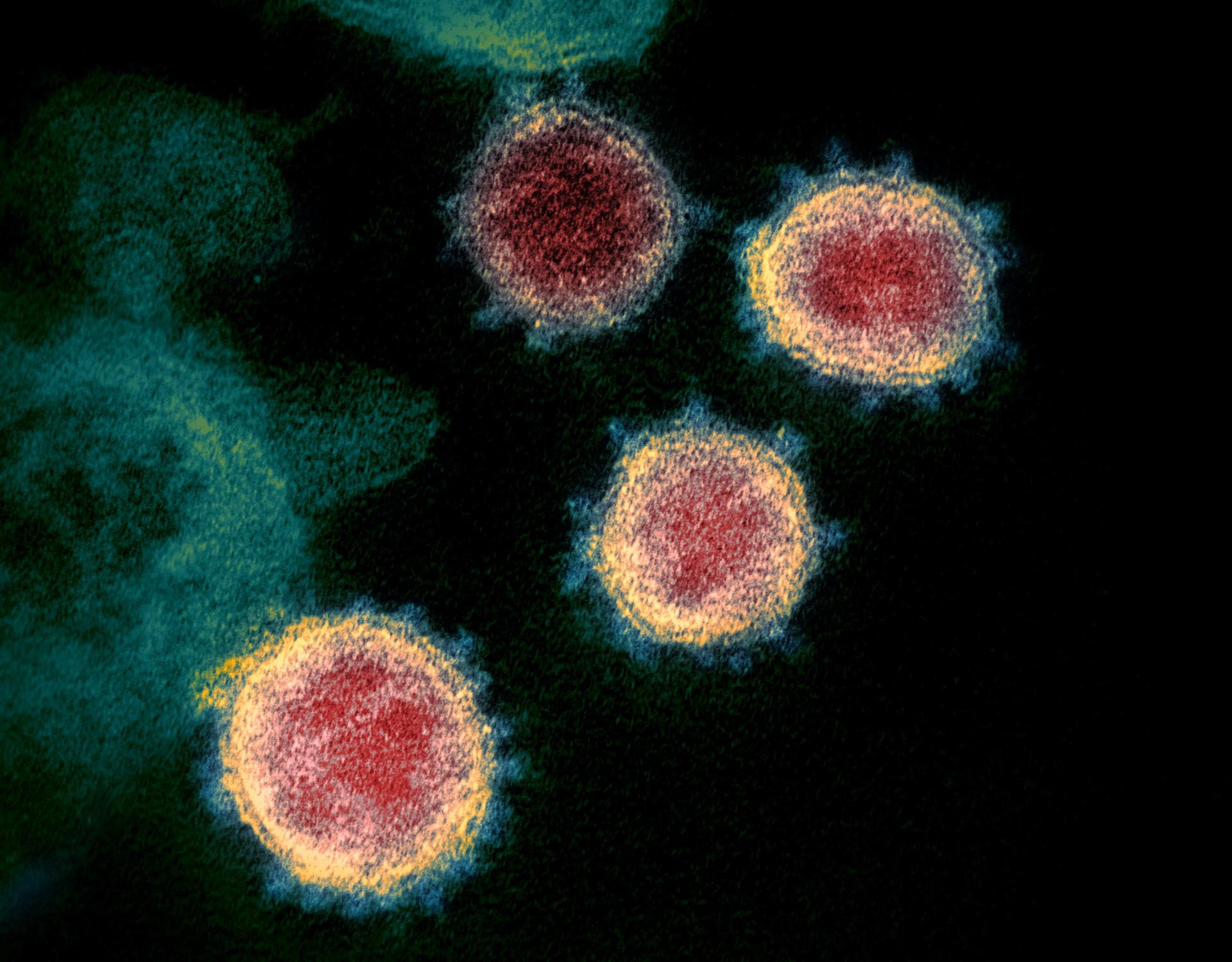
The SARS-CoV-2 variant, Omicron, shows rapid replication in human primary nasal epithelial cultures and efficiently uses the endosomal route of entry
At the end of 2021 a new SARS-CoV-2 variant, Omicron, emerged and quickly spread across the world. It has been demonstrated that Omicronâs high number of Spike mutations lead to partial immune evasion from even polyclonal antibody responses, allowing frequent re-infection and vaccine breakthroughs. However, it seems unlikely these antigenic differences alone explain its rapid growth; here we show Omicron replicates rapidly in human primary airway cultures, more so even than the previously dominant variant of concern, Delta. Omicron Spike continues to use human ACE2 as its primary receptor, to which it binds more strongly than other variants. Omicron Spike mediates enhanced entry into cells expressing several different animal ACE2s, including various domestic avian species, horseshoe bats and mice suggesting it has an increased propensity for reverse zoonosis and is more likely than previous variants to establish an animal reservoir of SARS-CoV-2. Unlike other SARS-CoV-2 variants, however, Omicron Spike has a diminished ability to induce syncytia formation. Furthermore, Omicron is capable of efficiently entering cells in a TMPRSS2-independent manner, via the endosomal route. We posit this enables Omicron to infect a greater number of cells in the respiratory epithelium, allowing it to be more infectious at lower exposure doses, and resulting in enhanced intrinsic transmissibility.