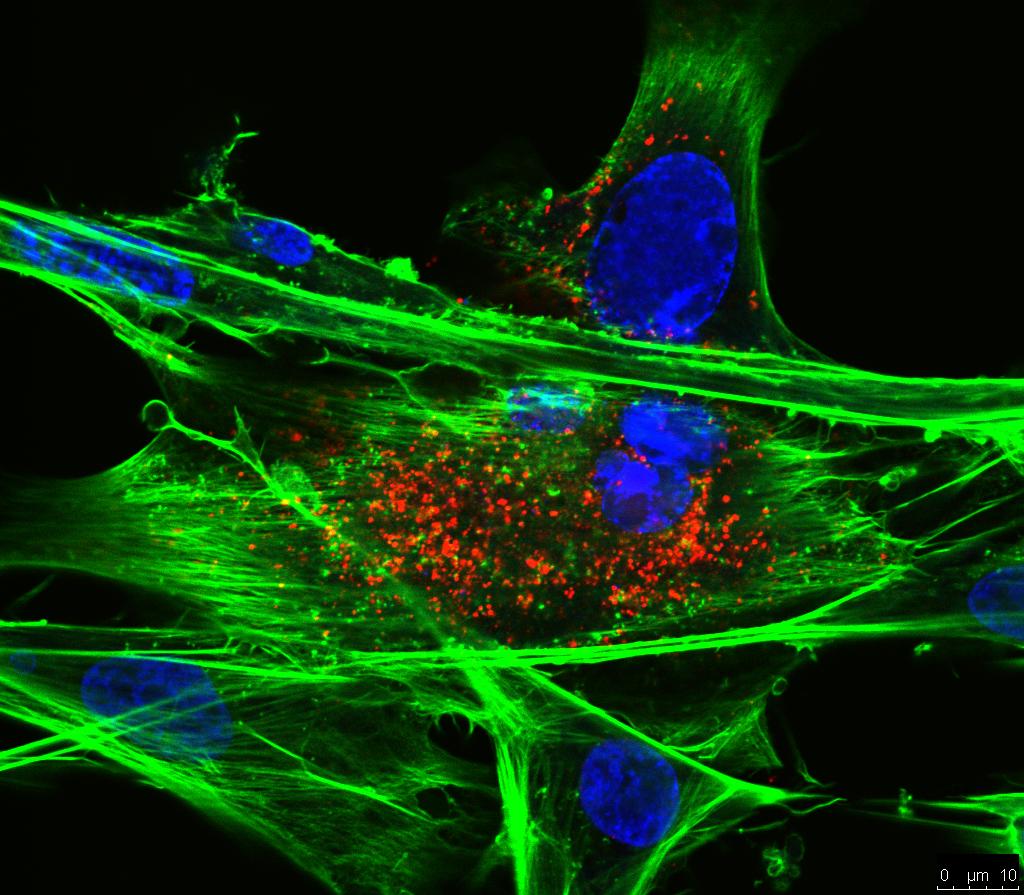
Glycosylation of ALV-J envelope protein at sites 17 and 193 is pivotal in the virus replication
Glycans on envelope glycoprotein (Env) of the subgroup J avian leukosis virus (ALV-J) play an essential role in virion integrity and infection process. In this study, we found that among the 13 predicted N-linked glycosylation sites (NGSs) in gp85 of Tibetan chicken strain TBC-J6, N17 and N193/N191 are pivotal in the virus replication. Further research illustrated that mutation at N193 weakened Env-receptor binding in blocking assay of viral entrance, co-immunoprecipitation and ELISA. Our studies also showed that N17 was involved in Env protein processing and later virion incorporation, based on the detection of p27 and Env protein in the supernatant and gp37 in the cell culture. This report is a systematic research on clarifying the biological function of NGSs on ALV-J gp85, which would provide valuable insights in the role of gp85 in ALV life cycle as well as anti-ALV-J strategies. Importance ALV-J is a retrovirus that can cause multiple types of tumors in chickens. Among all the viral proteins, the heavily glycosylated envelope protein is especially crucial. Glycosylation plays a major role in Env protein function, including protein processing, receptor attachment and immune evasion. Notably, viruses isolated recently seem to lose the 6(th) and 11(st) NGSs, which are proved to be important in receptor binding. In our study, the 1(st) (N17) and 8(th) (N193) NGS of gp85 of strain TBC-J6 can largely influence the titer of this virus. Deglycosylation at N193 weakened Env-receptor binding, while mutation at N17 influenced Env protein processing. This study systemically analyzed the function of NGSs in ALV-J in different aspects, which may help us to understand the lifecycle of ALV-J and provide antiviral targets for the control of ALV-J.