Our group
The research group was established in 2014 in the Division of Virology at the University of Cambridge with the support of a Marie Curie Fellowship to investigate molecular mechanisms of single stranded RNA virus protein translation. In 2016 we were awarded a Sir Henry Dale Fellowship from the Wellcome Trust and Royal Society, with an extension to the Fellowship awarded in 2021. In 2021 we moved to The Pirbright Institute to establish the Viral Gene Expression Group.
Our aims
We want to understand how viral RNA structures regulate interaction with viral and host factors to control different stages of the virus replication cycle. In this way we will uncover new details of viral replication that can be targeted for disease control.
Our research
Viruses cannot make their own proteins and instead must subvert the host protein synthesis pathway to replicate. We use a combination of in vitro reconstituted translation systems, protein-RNA interaction studies and cell-based experiments to study the molecular mechanism of protein synthesis, with a specific focus on how different viruses interfere with this critical cellular process.
RNA virus replication
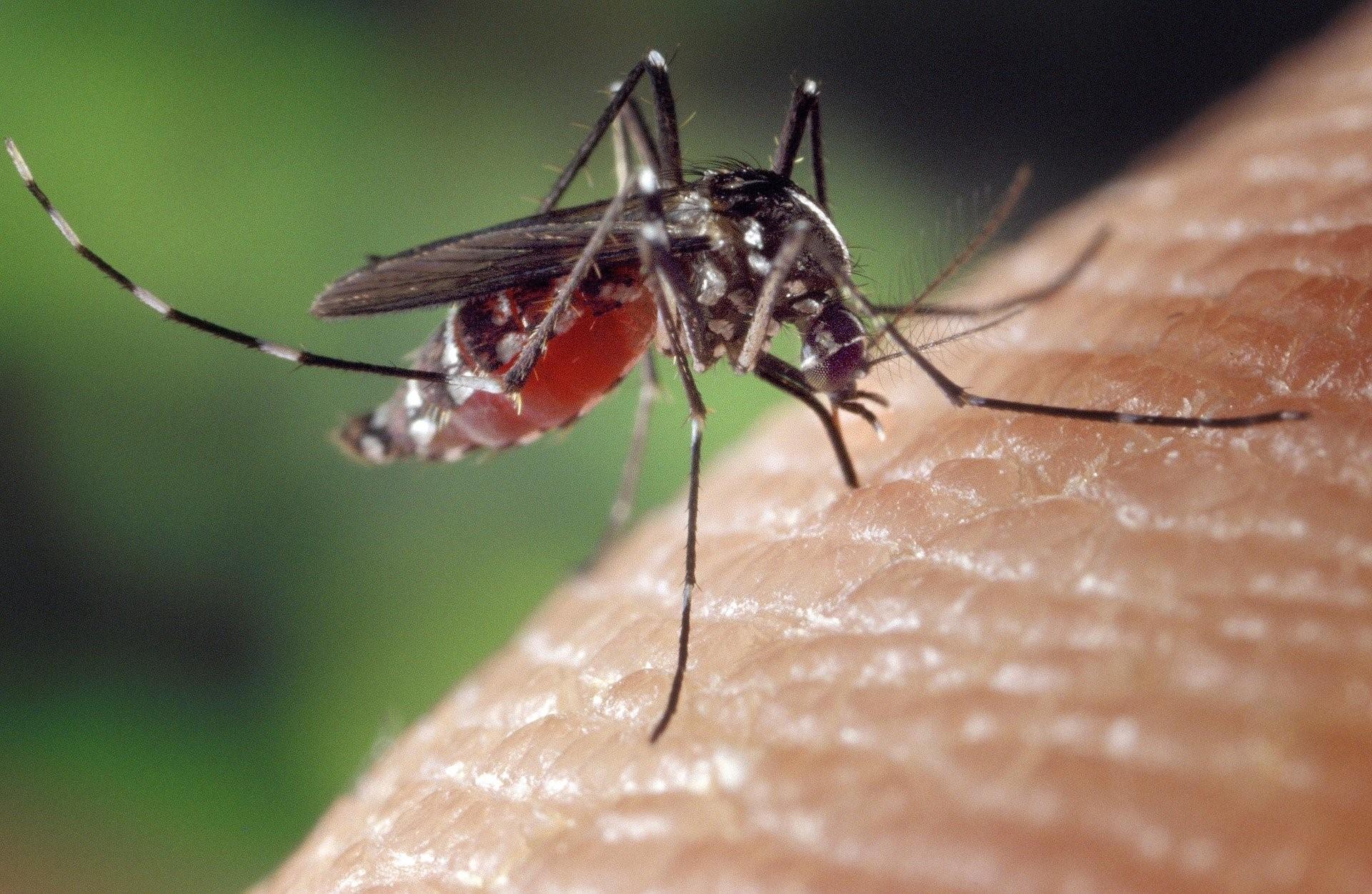
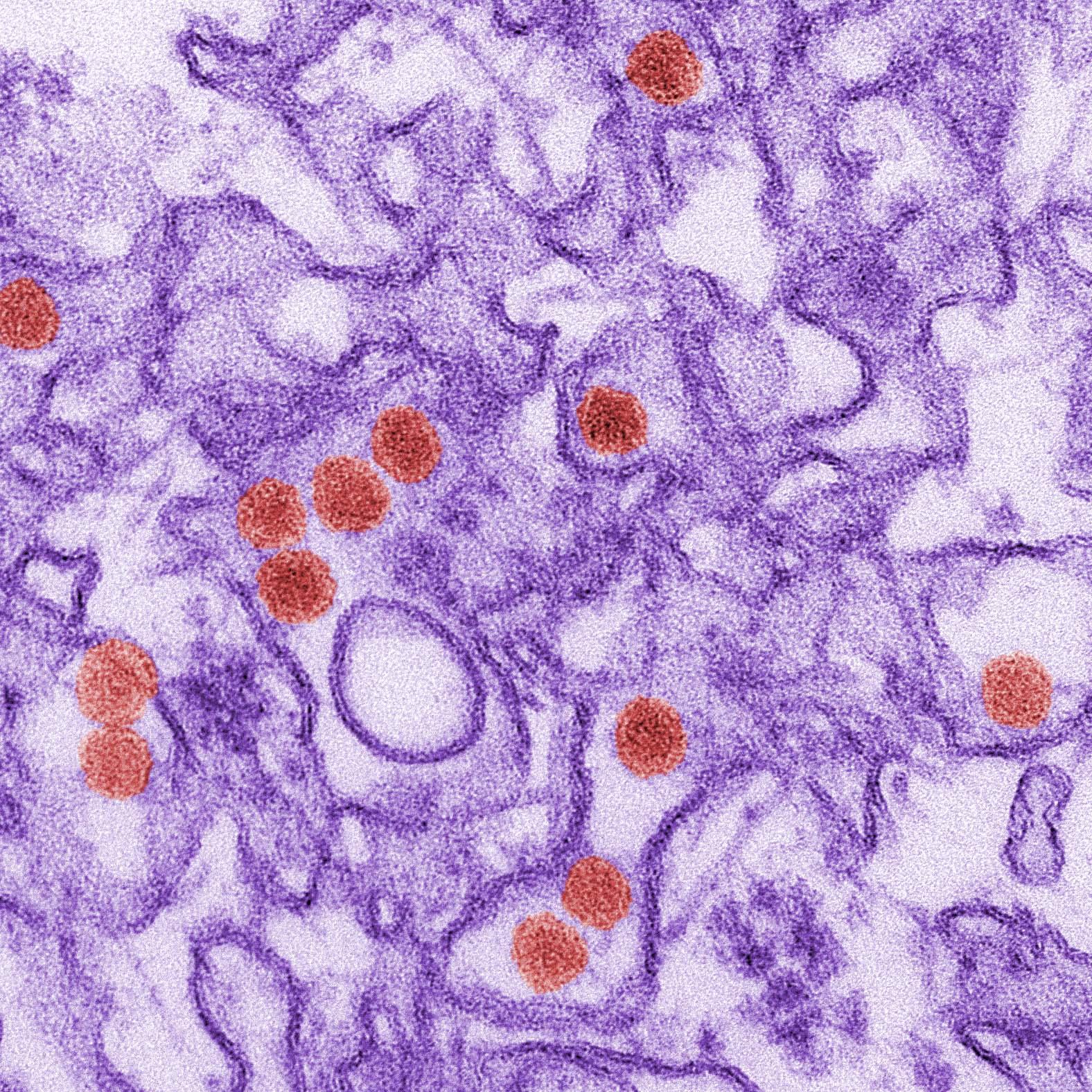
Flaviviruses such as West Nile virus, Zika virus and dengue virus have a single piece of RNA as their genetic material. This piece of RNA must be translated to make viral proteins and replicated to make new viruses. We are working to understand how these virus translate and replicate their genomes and regulate usage between these processes. This work is supported by the Wellcome Trust, the Royal Society and the BBSRC. Now based at The Pirbright Institute, we will expand our studies into other clinically important viruses such as foot-and-mouth disease virus, poliovirus and SARS-CoV2.
Immune regulation of virus translation
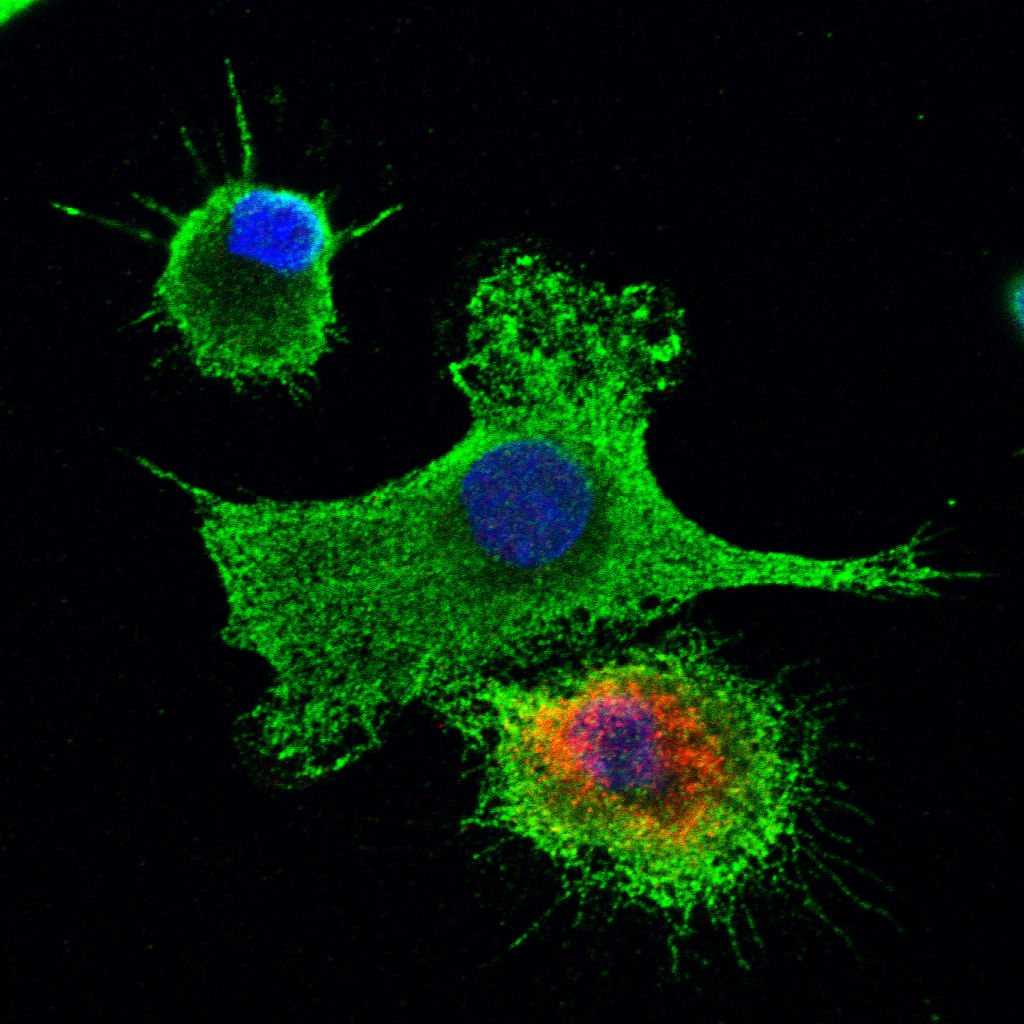
Another aspect of research in our lab is understanding the cellular response to infection and how the immune system specifically inhibits translation of viral RNAs. We characterized the mechanism of repression of viral translation by the interferon induced IFIT family of proteins. Binding of these proteins to specific 5′-RNA ends that are recognized as ‘non-self’ directly inhibited their translation. We are now examining the downstream effects off IFIT/RNA recognition and the role of oligomeric complex assemblies in regulating the cells response to infection. This work is supported by the Wellcome Trust, the Royal Society, the Newton Trust and the BBSRC.
Our impact
From foot-and-mouth-disease virus to Zika virus and SARS-CoV2, a number of threats to human and animal health are caused by viruses that use a single molecule of positive sense RNA as their genetic material. Expanding vector ranges and increased human encroachment on wildlife means we will continue experiencing outbreaks by this class of viruses. We have recently revealed new details of flavivirus (Zika virus, dengue virus) replication defining how changes in genome structure and interactions between viral proteins and viral RNA can affect viral protein synthesis and viral replication. Understanding how these viruses replicate in the cell will identify virus specific processes that can be rapidly targeted to help contain new outbreaks.