Our group
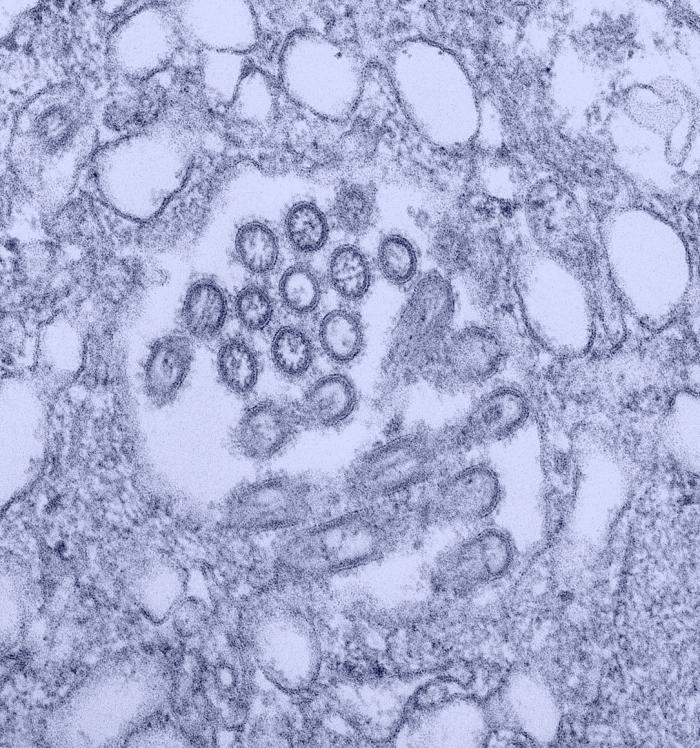
Respiratory viruses are amongst the greatest threats to global health.
There is an urgent need to better understand mechanisms of protective immunity to respiratory infections, such as influenza and coronaviruses, and to develop reliable animal models to test the efficacy of novel vaccines and therapeutics.
Because pigs are anatomically, physiologically, and immunologically more similar to humans than small laboratory animals, we have established a robust pig influenza challenge model to study influenza transmission and immunity and evaluate novel vaccines and therapeutics. More recently, we have developed a porcine respiratory coronavirus model with many similarities to human acute respiratory coronavirus disease to further understand infection with these high consequence viruses.
Our aims
The goal of our research is to develop better prevention strategies for respiratory pathogens in animals and humans. Our main objectives are:
- To determine the importance of local lung versus systemic immunity in protection against respiratory infections.
- To establish how to generate and maintain protective immunological memory against influenza and coronaviruses.
- To develop improved strategies to deliver mucosal vaccines and therapeutics to protect against different strains of respiratory viruses.
- To understand transmission dynamics of influenza and coronaviruses and identify means of preventing transmission.
Our research
We have developed powerful tools to study immunity in pigs, including cell transfer, tetramers, scintigraphy and means of enumerating tissue resident memory cells.
We have isolated the first pig influenza specific monoclonal antibodies, showing that they recognise identical parts of the virus to human antibodies.
These tools and experience make pigs an invaluable model to study immunity against respiratory infections for improvement of animal and human health.
We are currently studying:
- The key events and mediators that determine whether the outcome of influenza or coronavirus infection is mild or severe disease
- Dissecting the role of T cells and antibodies in the respiratory tract in broad protection against multiple strains of viruses
- The lifespan of tissue resident memory cells and the contribution of circulating cells to the tissue resident pool
- Blocking transmission by therapeutics acting either directly on the virus or the innate immune system
- Delivery of therapeutic monoclonal antibodies to uncover mechanisms underlying their ability to protect against disease, particularly the interactions between porcine IgG subclasses and Fc gamma receptors
- Isolating novel porcine monoclonal antibodies against influenza H3, H5, H7, hemagglutinin stem, and porcine coronavirus spike
Our impact
The pig is increasingly recognised as a powerful and reliable model for selection of vaccines and therapeutics for human influenza and coronaviruses.
- Improvement of vaccines targeting swine influenza will increase animal health, welfare and productivity in the pig industry by reducing severe disease and will lower costs for the consumer. Improved pathogen control reduces antibiotic treatment for secondary bacterial infections, the risk of contamination of the food chain and the environment and the development of antibiotic resistance.
- More efficient immunisation and transmission blocking strategies will reduce transmission and mitigate the zoonotic threat posed by swine influenza and coronaviruses.
- Development of novel vaccines and adjuvants that might be administered to the respiratory tract, offers the possibility of incorporating additional signals to alter memory generation and survival and may facilitate the development of long-lived cross-protective T cell memory against high consequence pathogens.