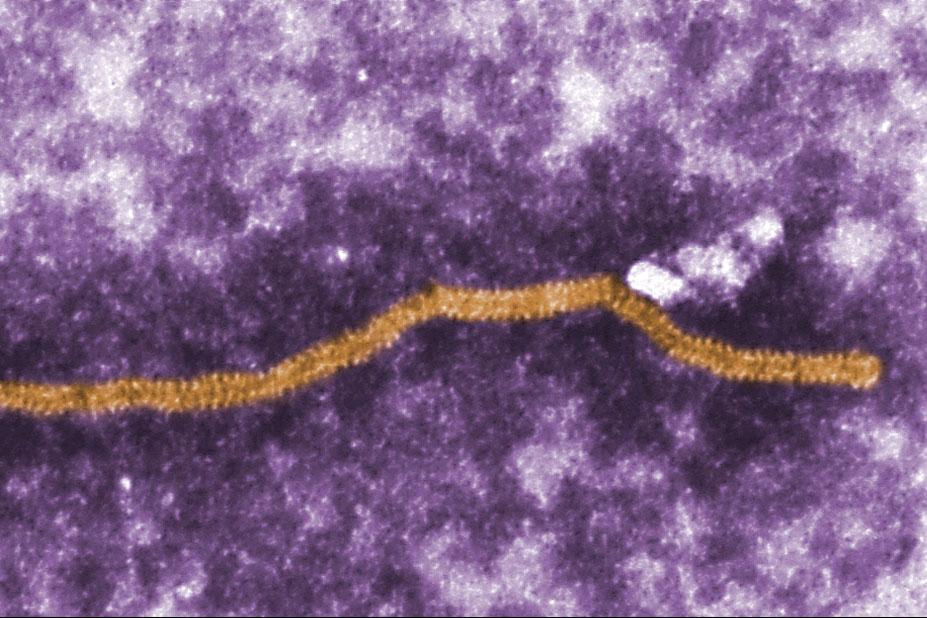
Nipah virus (NiV) is a newly emerging zoonotic infectious disease.
The natural hosts of NiV are fruit bats from the Pteropodidae family, specifically the Pteropus genus.
Pigs serve as amplifying hosts, facilitating easier transmission to humans.
There is evidence of Nipah infection among several other species of domestic animals including dogs, cats, goats, and horses.
NiV is transmitted through direct contact with the secretions and excretions of infected bats, pigs, and humans.
The disease first appeared in domestic pigs in Malaysia and Singapore in 1998 and 1999. Ten countries (Australia, Bangladesh, Cambodia, China, India, Indonesia, Madagascar, Papua New Guinea, Taiwan and Thailand) have reported Henipavirus (Nipah or the closely related Hendra virus) outbreaks, or are at risk based on serological evidence or molecular detection in Pteropus bats (CDC, USA, 2018).
Since the initial outbreak it has primarily affected humans in South and Southeast Asia (Malaysia, Singapore, Bangladesh and India) with devastating consequences.
Clinical signs
Pigs
- Respiratory and neurological syndrome, known as “barking pig syndrome”
- Morbidity is usually high but mortality is low
- Rapid laboured breathing
- Very harsh explosive cough
- In sows disease may be more pronounced with severe breathing difficulties
- Convulsions
- Pneumonia
- Mucopurulent discharges from the nose
- Death
Humans
Nipah virus infection in humans causes encephalitis (swelling of the brain).
The disease typically presents as fever and headache, followed by drowsiness and disorientation, which can rapidly progress to coma.
During the 1998-99 Nipah outbreak, 40% of patients admitted with serious nervous disease died. Respiratory signs may be observed during the early part of the infection, and a significant proportion of patients showing severe neurological signs also display pulmonary signs.
Latent Nipah virus infections have been reported with subsequent viral reactivation leading to death.
Virology
NiV is a RNA virus taxonomically classified within the family Paramyxoviridae, genus Henipavirus.
Pirbright's research on Nipah virus
Despite the huge threat that NiV poses, there are currently no vaccines available. Researchers at the Institute are therefore working on developing inexpensive, safe and effective vaccines to protect pigs, as well as diagnostic tests.
More broadly, we are also trying to understand whether pan-henipavirus vaccines can be an effective tool to prevent outbreaks of these viruses in susceptible animal and human populations.