Our group
We have a longstanding interest in understanding immunological mechanisms underlying protective immunity and the application of this knowledge to the development of new or improved vaccines.
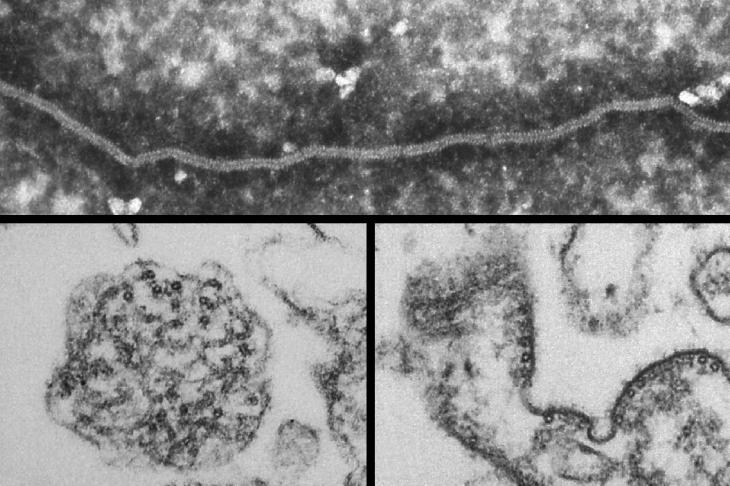
Our major research focus is the development of improved vaccines for porcine reproductive and respiratory syndrome (PRRS). PRRS is arguably one of the most important infectious disease of pigs with a huge global economic impact. The PRRS virus (PRRSV) exists as two distinct species: 1 and 2. Both PRRSV species are rapidly diversifying and this is dramatically illustrated by the emergence of highly pathogenic PRRSV-2 variants that spread across Southeast Asia with devastating effects. However, emerging PRRSV-1 strains are also highly virulent, highlighting the risks posed to the UK pig industry. Vaccination is a key element to PRRS control and while both inactivated and live attenuated PRRS vaccines are widely used neither provides sufficiently effective protection against the diversity of circulating PRRSV strains. There is therefore an urgent need to develop more efficacious vaccines to aid PRRSV control.
In addition, we are applying our expertise in porcine immunology and vaccinology to contribute to the development of vaccines to combat new and emerging zoonotic viruses, such as the Nipah virus and SARS-CoV-2.
Our aims
We are working with colleagues and collaborators on ambitious complimentary approaches aimed at the development of improved PRRS vaccines. We also aim to develop a safe and efficacious Nipah virus vaccine for pigs that will aid the prevention and control of Nipah outbreaks. And to support the development of COVID-19 vaccine candidates by utilising the pig as a preclinical model to study vaccine immunogenicity.
Our research
- Develop broadly cross-protective vaccines based on highly conserved neutralising antibody targets: We are deploying cutting edge technologies to identify and characterise naturally occurring porcine monoclonal antibodies capable of broadly neutralising PRRSV strains. These antibodies will then be used to identify highly conserved vaccine targets that may be engineered to induce cross-protection.
- Induction of effector T cell-based immunity by viral vectored delivery of conserved vaccine candidate antigens: Viral vector-based vaccines offer the potential to provide protection against virulent pathogens entering via the mucosal route. There is growing evidence that cell-mediated immune responses are critical for protection against PRRSV in the absence of neutralising antibodies. We have therefore identified and characterised conserved T cell antigens from PRRSV and are evaluating their vaccine potential using a novel vaccine vector platform based on an attenuated herpesvirus.
- Rational design of more efficacious live attenuated vaccines: We are evaluating a number of strategies to achieve this by:
- Identifying the key determinants of PRRSV proteins that mediate immune evasion or contribute to disease to provide rational targets for the production of attenuated vaccine strains.
- Evaluating whether a synthetic live attenuated PRRSV expressing ‘consensus sequence’ glycoproteins provides enhanced cross-protection.
- Genetically engineering PRRSV to remove decoy epitopes and glycan shielding or express peptide-based immune checkpoint inhibitors to promote the induction of neutralising antibody and T cell responses.
- Evaluating a novel solid dose vaccine formulation technology for delivery of PRRSV vaccines.
- Development of a Nipah virus vaccine to eliminate porcine reservoirs and safeguard human health. We are working with colleagues and partners to develop a prototype Nipah virus vaccine for pigs. We are evaluating a range of recombinant vaccine candidates, based on protein subunits as well as viral and mRNA vectors. We are also exploring the potential for live attenuated PRRSV and pseudorabies virus to act as vaccine vectors for Nipah and thus provide a bivalent vaccine.
- We are supporting the UK response to the COVID-19 pandemic by evaluating a range of vaccine candidates for immunogenicity in pigs to inform further clinical development.
Our impact
PRRSV is responsible for one of the most economically important infectious diseases affecting the global pig industry. PRRS control is hampered by the rapid evolution of the virus and shortcomings with existing vaccines. By dissecting the pathological and protective immune responses evoked by PRRSV, we will be able to design safer and more effective vaccines. We are working with global animal health businesses to develop improved vaccines that will contribute towards improving animal welfare and enhancing the performance and sustainability of the pig industry both in the UK and overseas.
Nipah virus causes a severe and often fatal disease in humans. Whilst fruit bats are considered the natural reservoir, Nipah virus also infects pigs and may cause an unapparent or mild disease. Direct pig-to-human transmission was responsible for the first and still most devastating Nipah outbreaks in Malaysia and Singapore in 1998-99. The outbreak was ended by culling 45% of the Malaysian pig population with costs exceeding US$500 million. The development of a vaccine to protect pigs would reduce the severe economic consequences of Nipah outbreaks and the threat to public health.
We were able to rapidly deploy the pig as a pre-clinical model to evaluate the immunogenicity of COVID-19 vaccine candidates. The vaccine candidates we have evaluated include the University of Oxford/AstraZeneca ChAdOx1 nCoV-19 vaccine, the University of Oxford’s virus-like particle displaying the SARS-CoV-2 S glycoprotein receptor-binding domain (RBD-SpyVLP), and Imperial College London’s lipid nanoparticle encapsulated self-amplifying RNA. The data generated have supported approvals for testing in clinical trials and, in the case of ChAdOx1 nCoV-19, supported successful Marketing Authorisation applications.